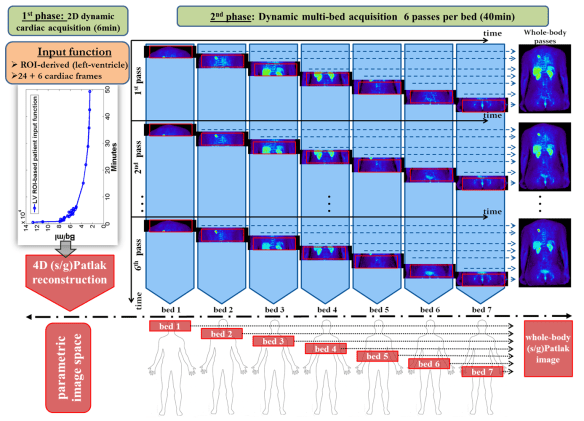
We are happy to introduce our latest method of 18F-NaF PET kinetics driven bone attenuation correction to enhance quantification of 18F-FDG and 18F-NaF images in clinical PET/MRI studies.
In our recent article at the Journal of Nuclear Cardiology, we present a robust method for segmenting bone tissues from dynamic PET data alone, thereby allowing for the direct attenuation correction of PET data from perfectly co-registered bone attenuation maps derived from the same PET data. This capability can be important for PET/MR studies where the gold-standard method for estimating attenuation maps from transmission data is not possible.
The bone tissue maps were segmented exclusively from parametric PET images which had been derived from robust Patlak analysis of the 18F-NaF kinetics. The remaining 4 basic tissue classes of air, lungs, fat and soft tissue can be segmented from state-of-the-art MR-based tissue segmentation methods. Finally, the 5 segmented tissue classes can be combined to build a unique 5-class hybrid PET/MR-driven attenuation map to more accurately correct for PET attenuation effects.

The novel hybrid PET/MR 5-class attenuation correction method has been designed to exploit the unique kinetic properties of 18F-NaF in standalone 18F-NaF as well as dual-tracer 18F-FDG:18F-NaF PET/MR studies to enable bone tissue segmentation exclusively from PET data. The figure below demonstrates the potential benefits of the method for improving contrast and quantification of both 18F-FDG and 18F-NaF carotid images acquired with a dynamic PET/MR acquisition protocol.
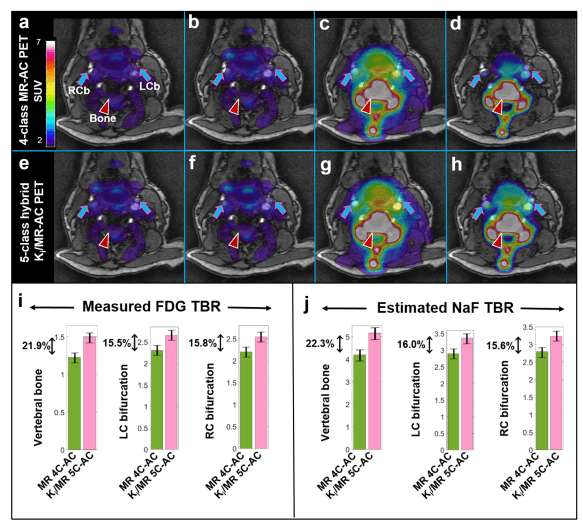
The new approach may present a robust and adoptable solution in PET/MR clinical studies to enhance PET signal quantification with two of the most widely employed PET radiotracers in cardiovascular and oncologic molecular imaging, that of 18F-FDG and 18F-NaF.